INTRODUCTION
The Pilbara Province of Western Australia, which extends from the De Grey River east of Port Hedland, to the Greenough River on the Geraldton Sandplains (Fig. 1), is one of ten freshwater fish biogeographic provinces in Australia (Unmack 2001; Unmack 2013). It is one of the most arid provinces. Annual rainfall is typically less than 400 mm, and streamflow mostly occurs in response to cyclonic activity or tropical lows in the summer wet season. However, some water persists where river channels intersect shallow aquifers, or gorges limit infiltration and evaporation (Masini 1988; Morgan et al. 2009; Pinder et al. 2010). These habitats provide critical refugia for 12 species of native, surface-dwelling freshwater fishes, with representatives from Terapontidae (grunters; three species), Atherinidae (hardyheads; one species), Melanotaeniidae (rainbowfishes; one species), Clupeidae (herrings; one species), Gobiidae (gobies; one species), Ariidae (fork-tail catfishes; one species), Plotosidae (eel-tail catfishes; one species), Anguillidae (freshwater eels; one species) and Eleotridae (gudgeons; two species) (Morgan & Gill 2004). Remarkably, the Pilbara Province also hosts three obligate subterranean fishes from Synbranchidae (swamp eels; one species) and Eleotridae (two species) (Morgan et al. 2014). Recent phylogenetic studies have shown that local representatives of Terapontidae, Plotosidae and Clupedidae likely represent cryptic species endemic to the region (Bostock et al. 2006; Morgan et al. 2014; Unmack 2013).
Among the Pilbara Province’s described freshwater fishes are two species from Hypseleotris (Eleotridae); the empire gudgeon Hypseleotris compressa Krefft, and the golden carp gudgeon Hypseleotris aurea Shipway (Morgan & Gill 2004). Hypseleotris compressa is distributed across southeastern and northern Australia, including the coastal reaches of most of the Pilbara Province’s major river systems (Fig. 1), as well as south-central Papua New Guinea (Shelley et al. 2023; Thacker & Unmack 2005) and New Britain Island off Papua New Guinea’s northeast coast (Keith & Mennesson 2023). Hypseleotris aurea is endemic to the Pilbara and mostly restricted to the Murchison and Gascoyne rivers in the province’s south (Fig. 1). An outlying Australian Museum record of H. aurea from 1981 also exists from a tributary of the Fortescue River (verified by Morgan & Gill 2004), which is over 200 km from the nearest H. aurea population (of the Gascoyne River) and separated by several major catchments (Fig. 1).
Here, we present phylogenetic analyses of specimens resembling Hypseleotris collected from a gorge pool in Karijini National Park (hereafter referred to as ‘the Karijini population’), approximately 15 km upstream of the 1981 Australian Museum record of H. aurea (Fig. 1). Due to its unique and isolated occurrence, our aim was to determine whether the Karijini population belongs to the genus Hypseleotris and, if so, to investigate its phylogenetic relationship to H. compressa and H. aurea, and other Hypseleotris from the Australasian region.
MATERIAL AND METHODS
Study material
Several specimens from the Karijini population were collected as bycatch in a macroinvertebrate sample during an aquatic ecological survey in November 2024. Three were deceased in the sample, which were placed in an ice slurry and then preserved in 100% ethanol. Additional live specimens were photographed in a small viewing tank (Fig. 2), before being returned alive to the capture site. The ethanol-preserved specimens were stored in a freezer prior to analysis. One ethanol-preserved Hypseleotris aurea specimen previously collected from the Gascoyne River near Yinnetharra Station was included in the analysis for comparative purposes.
Molecular analysis
DNA was extracted from dorsal fin clippings of the preserved fish using the QIAGEN DNeasy Blood and Tissue extraction Kit at the laboratory of Biologic Environmental, Western Australia. Two primer sets (GOBY L4919 and GOBY H5513; HYPSL5464 and GOBY H6064; Thacker & Unmack 2005) were used to amplify overlapping segments of the mitochondrial ND2 (NADH dehydrogenase subunit 2) gene. Amplified PCR products were sequenced using bidirectional Sanger sequencing at the Australian Genomic Research Facility (AGRF), Perth. Raw sequence data were edited and assembled in GENEIOUS Prime (Kearse et al. 2012), and the final consensus sequences were compared against the GenBank database (https://www.ncbi.nlm.nih.gov/genbank/), using the “blastn” function. Reference sequences of other Hypseleotris species were retrieved from the GenBank nucleotide database (Table 1). Additional ND2 sequences were derived from UCE-enriched genomic reads deposited in the NCBI SRA repository under BioProject PRJNA774634, kindly provided by Christine E. Thacker (Table 1).
The selected sequences were aligned using the Multiple Alignment using Fast Fourier Transform (MAFFT) algorithm (Katoh et al. 2002). Phylogenetic trees were reconstructed using two approaches: Maximum Likelihood (ML) and Bayesian Inference (BI). The ML tree was generated with the RaxML (Stamatakis 2014) plugin in GENEIOUS Prime, using 1,000 bootstrap replicates and the GTR+G substitution model. BI was performed in BEAST 1.10.5 (Suchard et al. 2018), using Markov chain Monte Carlo (MCMC) sampling every 1,000 generations over a total of 10 million generations. The maximum clade credibility tree (posterior probability limit set to 0.95) for BI was reconstructed in TreeAnnotator. The final phylogenetic tree images obtained from ML and BI were visualised in FigTree v.1.4.3 (http://tree.bio.ed.ac.uk/software/figtree).
Since the ML and BI analysis generated congruent topologies, only the BI tree is presented here, with both ML bootstrap values (BS) and BI posterior probabilities (BPP) indicated at the nodes. Uncorrected genetic distances (p-distances) were calculated in GENEIOUS Prime. The sequences were deposited in GenBank (Accession Numbers PX619673, PX619674, PX619675 and PX619676).
RESULTS
Habitat description
The habitat of the Karijini population comprised a narrow (<5 m), moderately deep (>1.5 m) pool confined within a steep, amphitheatre-shaped gorge. A series of stepped, rocky ledges covered in a thick biofilm/algal layer were present on the pool’s margins, before a steep drop off in the centre. No aquatic macrophytes, woody debris or other in-stream micro-habitats were present. The target fish were abundant in the pool (over 200 specimens observed), and favoured the top third of the water column, mostly hovering above the peripheral rocky ledge. No other fish species were observed at the time of collection.
The collection site is situated at the headwaters of a short (<30 km long) tributary of the Fortescue River. Between wet season flow events, numerous disconnected pools persist where bedrock and cliff faces would limit infiltration and evaporation. The system drains in a north-easterly and then northerly direction through a series of gorges, before emptying via alluvial fan into the central Fortescue Valley floodplain; an area containing a mosaic of interconnected, highly ephemeral claypans. It is likely the system rarely connects with the Fortescue River main channel, except during very large flooding events.
Phylogenetic analysis
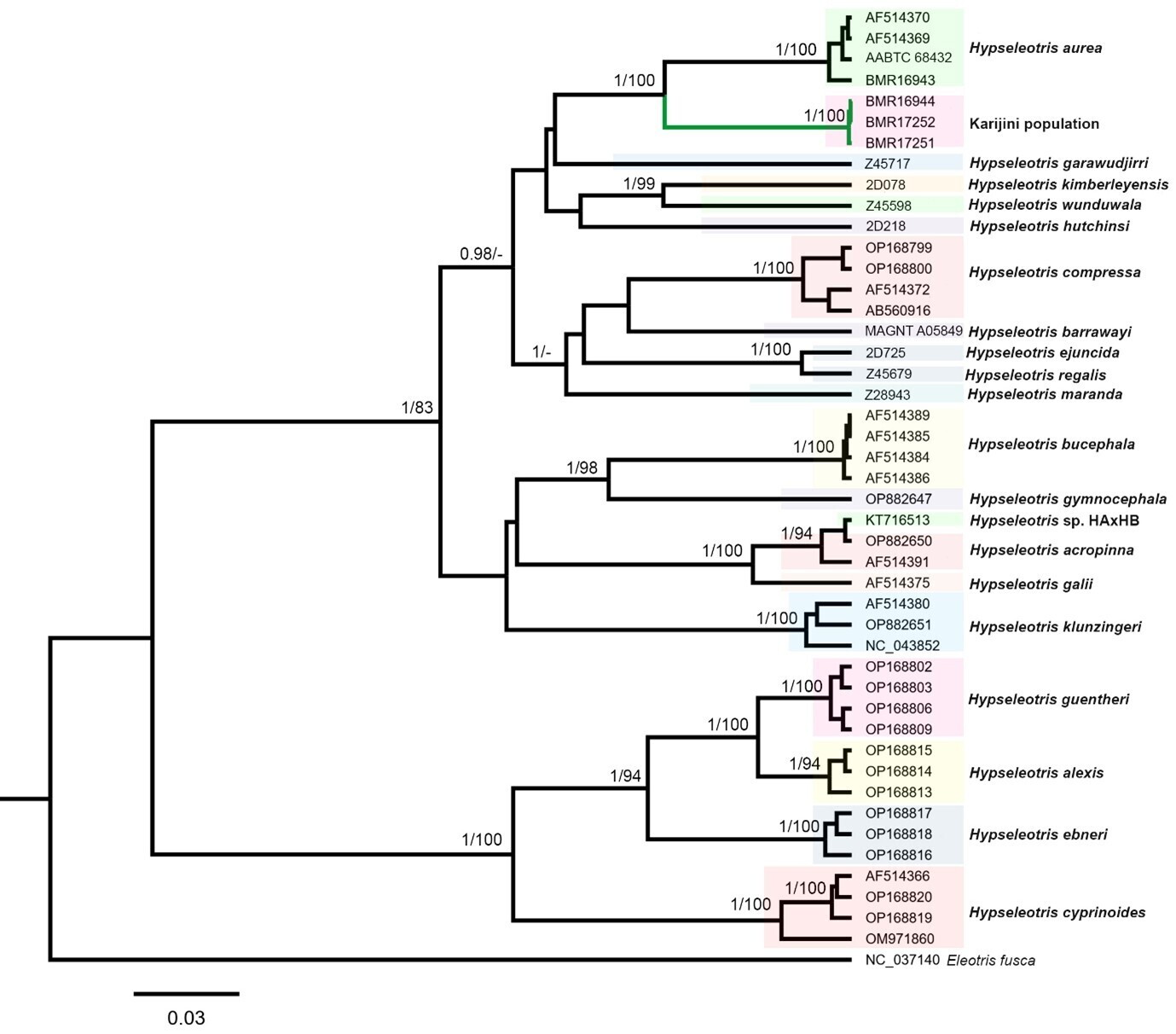
The mitochondrial ND2 gene segment (1,047 bp) was successfully amplified from three specimens from the Karijini population (BMR16944, BMR17252, BMR17251) and the H. aurea specimen from the Gascoyne River (BMR16943). Phylogenetic analyses of the ND2 dataset, including other Hypseleotris sequences, yielded congruent topologies in both BI and ML, with the Karijini population forming a distinct and strongly supported monophyletic lineage within the Hypseleotris genus (BPP = 1.0, BS = 100; Fig. 3). The closest lineage to the Karijini population in the phylogenetic tree was H. aurea with strong support (BPP = 1.0, BS = 100; Fig. 3). The Karijini population was more than 8.6% divergent from H. aurea, and between 11.6–18.9% divergent from other Hypseleotris species (Table 2).
DISCUSSION
The Karijini population represents a distinct lineage within Hypseleotris, forming a well-supported clade sister to H. aurea (Fig. 3). The lowest divergence between the Karijini population and H. aurea (8.7%) exceeded that between several recognised congeners in the analysis. For example, H. hutchinsi was 8.5–10.0% divergent from H. compressa, H. ejuncida differed by only 2.5% from H. regalis, and H. galii by 4.1–4.4% from H. acropinna (Table 2). Its minimum divergence from H. aurea was also greater than that reported between various Hypseleotris species from Indo-Pacific islands, including H. ebneri and H. guentheri (8.0–8.4%), H. ebneri and H. alexis (7.7–8.5%), and H. guentheri and H. alexis (4.0–4.8%) (Keith & Mennesson 2023). These comparatively low interspecific distances among other Hypseleotris species highlight the clear genetic distinction of the Karijini population from H. aurea (and other congeners), providing preliminary support for its recognition as a separate species.
Besides the 1981 Australian Museum record of H. aurea, which we consider likely belongs to the Karijini population given its occurrence on the same tributary, and H. compressa, which has a coastal distribution, there are no verified Hypseleotris records from the Pilbara Province outside of the Gascoyne and Murchison rivers. This is despite a relatively recent, extensive regional ichthyological survey (Morgan & Gill 2004), and numerous aquatic ecological impact assessment studies for mining projects by various consultancy firms (e.g. Biologic Environmental, Stantec Australia, WRM, Streamtec; see Appendix Table 1). As such, there is a strong possibility that the Karijini population is restricted to the 15 km stretch of tributary between the Australian Museum record and our collection location. Many Hypseleotris of northwest Australia are range-restricted, and it is not uncommon for species to be endemic to one catchment or sub-catchment, or even single creeks or rivers (Morgan & Gill 2004; Shelley et al. 2018; Thacker & Unmack 2005; Unmack 2001). Examples from the nearby Kimberley Province include H. wunduwala, limited to three locations on the Carson River, a tributary of the King Edward River, and H. maranda which only occurs at Garimbu Creek within the Roe River catchment (Shelley et al. 2023). It is likely that the Karijini population and H. aurea once shared a common, more widespread ancestor, with populations becoming isolated in response to acidification during the late Miocene and Pleistocene, as has been postulated for other range-restricted Hypseleotris in northwest Australia (Thacker & Unmack 2005). Presently, the highly ephemeral Fortescue Valley floodplain at the downstream terminus of its home tributary may prevent dispersal into the broader Fortescue River catchment, while sheltering the population from predatory fishes.
Our results add to the growing body of genetic evidence that there are likely to be more freshwater fishes in the Pilbara Province than the 15 formally described. Bostock et al. (2006) found evidence for two cryptic species among the province’s endemic Fortescue grunters (Leiopotherapon aheneus), while the Pilbara’s bony bream (Nematolosa erebi) and Hyrtl’s tandan (Neosilurus hyrtlii) are also genetically distinct from those of neighbouring provinces, each likely representing an undescribed endemic species (Morgan et al. 2014; Unmack 2013). More broadly, Hammer et al. (2013) proposed there may be twice as many freshwater fishes in Australia than currently recognised, due to cryptic speciation. Against the backdrop of an expansive mining industry, for which the Pilbara is renowned (Morgan et al. 2014), and increasing reports of invasive aquatic species, with sailfin molly (Poecilia latipinna) and redclaw crayfish (Cherax quadricarinatus) being recently introduced to the Fortescue River catchment (Pinder et al. 2019; Thorburn et al. 2018), there is a need for more frequent, robust phylogenetic studies to aid conservation planning for the Pilbara Province’s freshwater fishes. To this end, further genetic work is required to untangle the phylogeny of the Pilbara Province’s various Hypseleotris populations. There are few publicly available sequences for H. aurea of the Gascoyne River (limited to those used in this study), and we could not source any genetic information for H. aurea of the Murchison River. Detailed morphological work would also be required to investigate differences between the populations and potentially aid in the description of the Karijini population as a new species.
CONCLUSIONS
The Karijini population, potentially restricted to a single tributary of the Fortescue River, represents a distinct genetic lineage within Hypseleotris. Its minimum divergence from sister species H. aurea is greater than that recorded between several described Hypseleotris species from northwest Australia and the Indo-Pacific region, providing strong support for recognition as a separate species, though further genetic and morphological work is required to untangle its phylogeny. Our results add to the increasing number of putative freshwater fishes being reported in the Pilbara province based on genetic evidence. Against the backdrop of an expansive mining industry and increasing spread of invasive species, there is a growing need for further genetic work to aid conservation planning for the Pilbara Province’s ichthyofauna.
ACKNOWLEDGEMENTS
This study was funded by Biologic Environmental (Biologic). Several of our colleagues at Biologic are thanked for their contributions, particularly Shriya Bhattacharya who performed specimen subsampling and laboratory analysis, Diana Brittain who created the map, and Jess Delaney for reviewing the draft manuscript. Thanks also to Suzi Wild (BHP) and the Karijini National Park rangers for their endorsement of the study. We are grateful to Dr. Christine E. Thacker for providing sequences of Hypseleotris species collected from Western Australia. The specimens used in this study were collected under the following licences and permits: Fauna Taking (Biological Assessment) Licences (Regulation 27) BA27000421-4 and BA27001076, issued by the Western Australian Department of Biodiversity, Conservation and Attractions (DBCA); Regulation 4 Authority-8(1) CE006993 issued by DBCA; Instrument of Exemption to the Fish Resources Management Act – Section 7(2) exemption numbers EXEM250976722 and EXEM251252424, issued by the Western Australian Department of Primary Industries and Regional Development (DPIRD); and Licence to use animals for scientific purposes U244/ 2022-2024, administered through DPIRD.
.jpg)

.jpg)